Why Structured Testing Workflows Are Essential in Autoimmune Diagnostics
Structured workflows in autoimmune diagnostics from ANA screening to ENA profiling helping laboratories improve diagnostic clarity, operational efficiency, and result reliability.
Key Takeaways
- Autoimmune diagnostics requires structured testing workflows, as individual assays alone rarely provide sufficient diagnostic clarity.
- ANA screening tests are widely used as the first step in autoimmune evaluation, but positive results require further investigation because ANA antibodies can also appear in healthy individuals.
- Autoantibody profiling tests such as ENA panels help identify disease-specific antibody patterns, enabling laboratories to differentiate between autoimmune disorders.
- Different immunoassay technologies—including IFA, ELISA, and LIA—play complementary roles in autoimmune diagnostic workflows by balancing sensitivity and specificity.
- Structured diagnostic pathways improve laboratory efficiency, helping reduce unnecessary testing, repeat runs, and interpretational uncertainty.
- Consistent assay performance across screening and profiling stages is essential for maintaining diagnostic confidence in autoimmune testing.
Why Autoimmune Diagnostics Requires Structured Testing Workflows
On a typical morning in a diagnostic laboratory, a sample arrives with a request that laboratories see every day:
“Rule out autoimmune disease.”
The patient may present with persistent fatigue, joint pain, unexplained inflammation, or systemic symptoms that do not clearly point toward a single diagnosis. For clinicians, autoimmune diseases often represent a diagnostic puzzle. For laboratories, they represent a diagnostic journey rather than a single test result.
Autoimmune conditions rarely reveal themselves through one assay alone. Instead, accurate diagnosis emerges through a sequence of investigations that gradually transform early immune signals into clinically meaningful insights. In modern laboratories, this transformation relies on structured diagnostic workflows that move from screening to profiling and targeted investigation.
Understanding this structure is essential for laboratories aiming to deliver reliable autoimmune diagnostics.
The Diagnostic Complexity of Autoimmune Diseases
Autoimmune diseases arise when the immune system mistakenly produces antibodies against the body’s own tissues. These autoantibodies can target a wide range of cellular components including nuclear proteins, cytoplasmic antigens, and connective tissue structures.
Conditions such as:
- Systemic lupus erythematosus (SLE)
- Sjögren’s syndrome
- Systemic sclerosis
- Mixed connective tissue disease
often develop gradually and present with symptoms that overlap with other inflammatory or infectious disorders.
For clinicians, diagnosis is rarely immediate.
For laboratories, this means autoimmune testing must be structured and sequential, allowing early immune signals to be interpreted within a broader diagnostic framework.
The Challenge of ANA Positivity Without Disease
One of the most widely used screening tests in autoimmune diagnostics is the antinuclear antibody (ANA) assay.
ANA testing is highly sensitive for many systemic autoimmune diseases and serves as an effective first-line screening tool. However, ANA positivity does not automatically indicate disease.
Population studies show that approximately 12–16% of healthy individuals may demonstrate ANA positivity, particularly at lower antibody titers, depending on assay methodology and thresholds.
(Source: https://www.mdpi.com/1648-9144/61/1/60)
Other research has reported ANA antibodies in up to about 25% of individuals depending on testing sensitivity and population characteristics, although only a small proportion of these individuals will develop autoimmune disease.
(Source: https://pmc.ncbi.nlm.nih.gov/articles/PMC3132017/)
Age, infections, medications, and transient immune responses may also contribute to ANA positivity.
For laboratories, this creates a diagnostic challenge.
A positive ANA result signals immune activity but does not explain its clinical significance. Without careful follow-up testing and interpretation, ANA results can lead to diagnostic uncertainty rather than clarity.
This reinforces the importance of structured diagnostic pathways that extend beyond screening.
The Role of ANA Screening in Autoimmune Diagnostics
Returning to our laboratory scenario, the first diagnostic step typically involves screening for autoimmune activity using ANA testing.
Common screening methodologies include:
- Indirect Immunofluorescence Assay (IFA)
- ELISA-based ANA screening
These assays allow laboratories to detect antibodies directed against nuclear components of the cell.
When ANA results are negative, systemic autoimmune disease becomes less likely. When they are positive, however, laboratories have identified a signal—but not yet a diagnosis.
At this stage, the diagnostic process has only begun.
Clinical guidelines emphasize that ANA testing plays an important role in the evaluation and classification of systemic autoimmune diseases, but results must always be interpreted alongside clinical findings and additional laboratory tests.
(Source: https://myadlm.org/cln/articles/2019/april/a-basic-guide-to-antinuclear-antibody-ana-testing)
Bridging the Gap Between Detection and Diagnosis
A positive ANA result often raises more questions than it answers.
Clinicians may suspect autoimmune disease, but ANA positivity can occur across multiple conditions and sometimes in healthy individuals. Without additional testing, the laboratory report may offer limited diagnostic direction.
This is where structured workflows become critical.
Rather than stopping at screening results, experienced laboratories proceed to autoantibody profiling assays designed to identify specific antibodies associated with particular autoimmune diseases.
These profiling tests transform broad immune signals into clinically interpretable patterns.
Autoantibody Profiling and ENA Panels
Autoantibody profiling panels—such as Extractable Nuclear Antigen (ENA) panels—detect antibodies directed against specific nuclear proteins.
These include markers such as:
- Anti-Ro (SSA)
- Anti-La (SSB)
- Anti-Sm
- Anti-RNP
- Anti-Scl-70
- Anti-Jo-1
Each of these antibodies is associated with different autoimmune conditions.
For example:
- Anti-Sm antibodies are strongly associated with systemic lupus erythematosus.
- Anti-Ro and Anti-La antibodies are commonly linked to Sjögren’s syndrome.
- Anti-Scl-70 antibodies may indicate systemic sclerosis.
ENA testing is often recommended after positive ANA screening to help identify disease-specific antibody patterns and support clinical diagnosis.
(Source: https://www.testing.com/tests/extractable-nuclear-antigen-antibodies-ena-panel/)
By identifying these patterns, profiling assays provide clinicians with crucial diagnostic clues that help differentiate between autoimmune diseases.
The laboratory moves from detection to characterization.
Different Technologies in Autoimmune Testing (ELISA, IFA, LIA)
Modern autoimmune diagnostics relies on several immunoassay technologies, each serving a distinct purpose within the testing workflow.
Indirect Immunofluorescence (IFA)
Often regarded as a highly sensitive screening method, IFA allows visualization of antibody binding patterns within cells.
ELISA-based assays
ELISA tests provide automated, high-throughput screening capabilities and are widely used in routine laboratory workflows.
Line Immunoassays (LIA)
LIA platforms allow simultaneous detection of multiple autoantibodies, enabling efficient profiling of autoimmune markers.
Studies evaluating ANA detection technologies highlight that these testing methods serve complementary roles within structured diagnostic algorithms, balancing sensitivity and specificity across testing stages.
(Source: https://pmc.ncbi.nlm.nih.gov/articles/PMC2628865/)
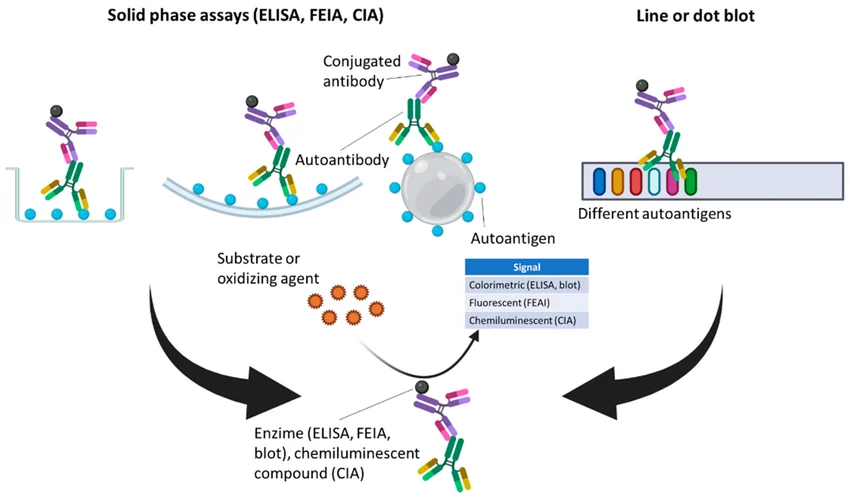
Figure: Complementary roles of immunoassay technologies in autoimmune diagnostics
Different immunoassay technologies contribute unique strengths to autoimmune diagnostics. Indirect immunofluorescence assays are widely used for sensitive ANA screening. ELISA-based assays enable high-throughput automated screening in routine laboratories. Line immunoassays allow multiplex detection of multiple autoantibodies simultaneously, supporting detailed antibody profiling. When integrated into structured diagnostic workflows, these technologies provide both sensitivity and specificity.
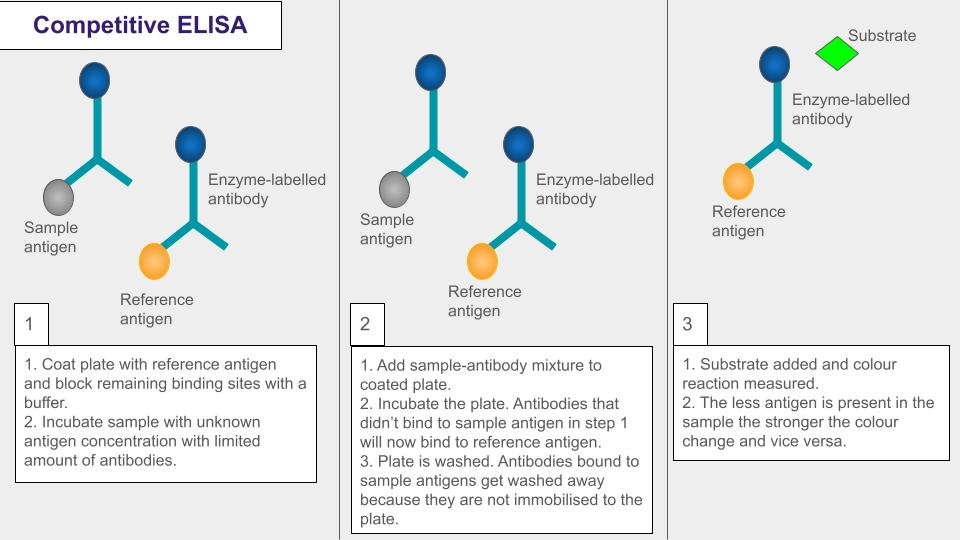
Source: https://www.whatisbiotechnology.org/index.php/science/summary/immunoassays?utm_source=chatgpt.com
Diagram illustrating how the competitive ELISA works. Adapted from Mészáros. The competitive ELISA method is often used to measure the concentrations of small molecules and hormones.
Source: https://www.whatisbiotechnology.org/index.php/science/summary/immunoassays?utm_source=chatgpt.com
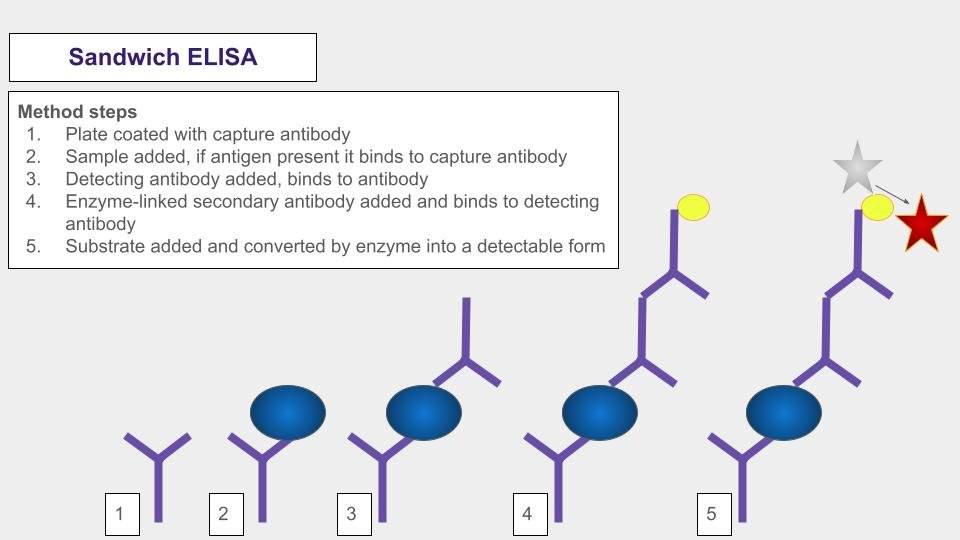
Diagram of a sandwich assay adapted from Jeffrey M Vicour, Wikipedia. Sandwich ELISAs are now one of the most common types of ELISA method used. Two antibodies are used in the test to bind to the protein of interest which makes it a highly specific test.

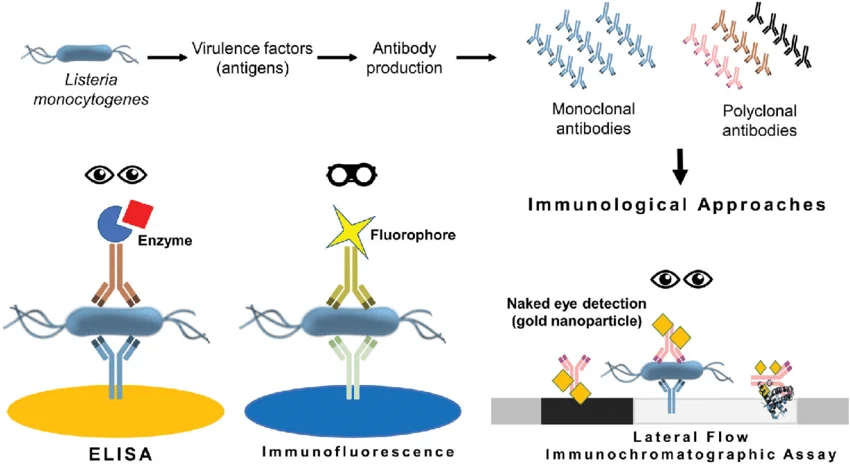
Immunoassay formats include ELISA (Enzyme-Linked Immunosorbent Assay), Immunofluorescence Assay, and Lateral Flow Immunochromatographic Assay.
From Individual Tests to Diagnostic Workflows
In advanced diagnostic laboratories, autoimmune testing is rarely performed as isolated assays. Instead, laboratories design structured diagnostic pathways that guide testing from screening to disease-specific profiling.
A typical workflow may include:
Screening → Profiling → Targeted Investigation
This approach offers several advantages:
- clearer diagnostic interpretation
- reduced unnecessary testing
- more efficient reagent usage
- consistent reporting for clinicians
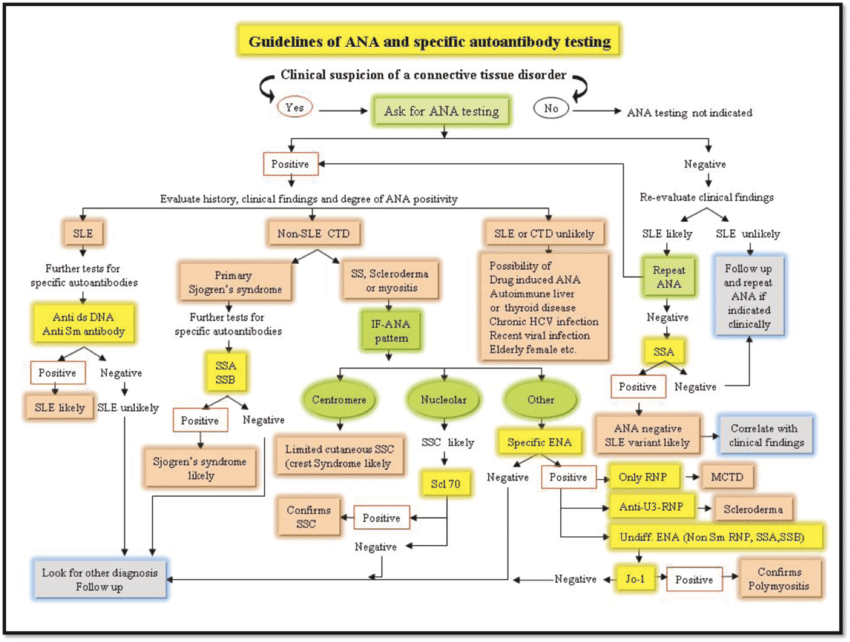
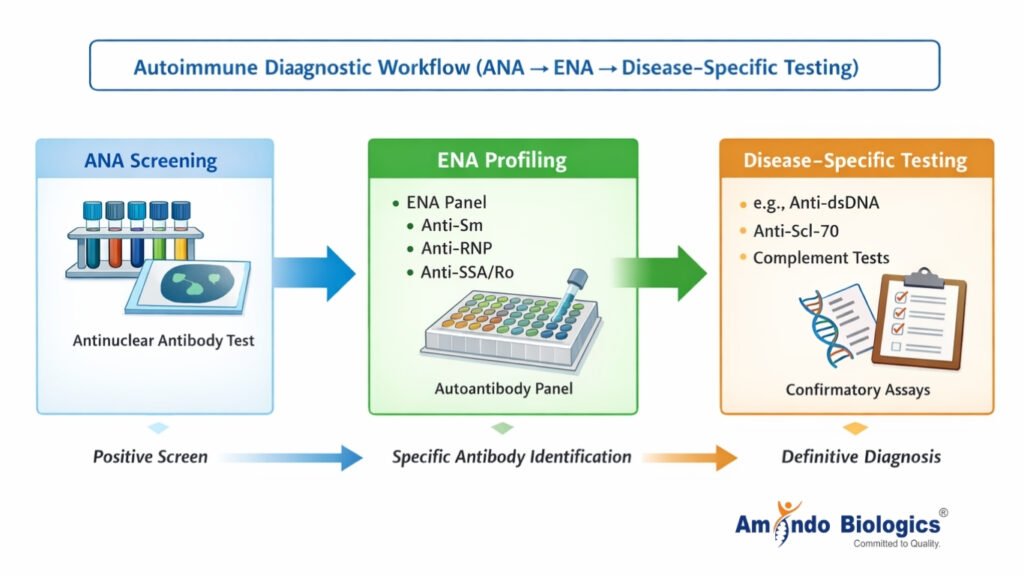
Source: https://www.researchgate.net/figure/Algorithmic-approach-for-ANA-testing_fig1_283682717
Figure: Structured autoimmune diagnostic workflow
Most laboratories follow a sequential testing approach when evaluating suspected autoimmune disease. Screening assays such as ANA tests identify the presence of autoantibodies. When screening results are positive, laboratories typically proceed to profiling assays such as ENA panels to identify disease-specific antibodies. In some cases, additional targeted testing is performed to confirm diagnosis or monitor disease progression.
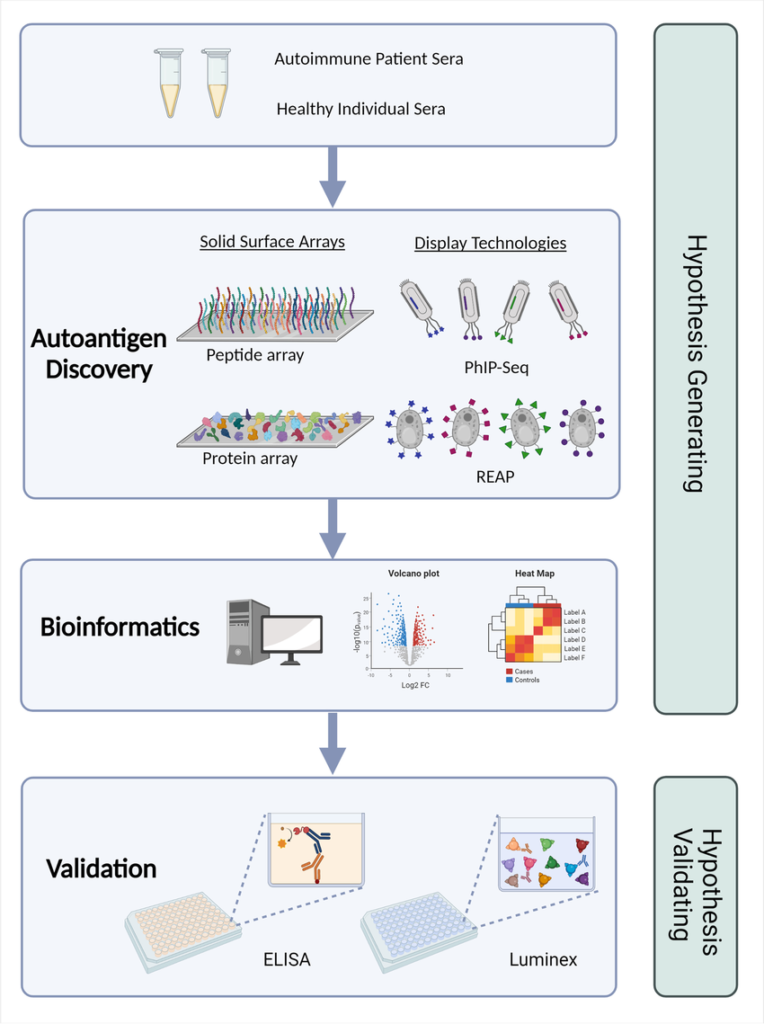
Structured workflows allow laboratories to transform complex immune signals into coherent diagnostic narratives.
Operational Efficiency in Autoimmune Diagnostic Workflows
Behind every autoimmune diagnostic result lies a complex laboratory process.
Technologists ensure assays perform consistently across batches. Laboratory managers maintain workflow efficiency while handling increasing testing volumes. Pathologists interpret results in conjunction with clinical data.
When diagnostic systems function smoothly, laboratories deliver results efficiently.
However, when inconsistencies arise—whether in assay performance, workflow processes, or reagent reliability—laboratories may begin to experience operational challenges such as:
- repeat testing
- delayed reporting
- increased reagent consumption
- interpretational uncertainty
These issues rarely appear suddenly. Instead, they often emerge gradually as diagnostic predictability begins to decline.
Why Consistency Matters in Autoimmune Diagnostics
Because autoimmune testing integrates multiple assays within a single diagnostic pathway, laboratories depend heavily on consistent performance across screening and profiling stages.
Variations in reagent stability, assay reproducibility, or batch consistency can affect diagnostic interpretation when results from multiple tests must be combined.
For laboratories, maintaining predictable assay performance across the entire diagnostic workflow is essential for sustaining confidence in results.
In autoimmune diagnostics, reliability is not determined by a single test but by the stability of the system that connects them.
The Future of Autoimmune Diagnostics
Advances in immunoassay technologies, laboratory automation, and multiplex testing platforms continue to improve the efficiency of autoimmune diagnostics.
Modern laboratories increasingly integrate automated screening systems with multiplex profiling panels to deliver faster and more comprehensive diagnostic insights.
Despite these technological advances, one principle remains fundamental:
Autoimmune diagnostics is not defined by individual assays, but by the logic that connects them.
Laboratories that design testing workflows around this principle are better equipped to navigate the complexity of immune-mediated diseases.
Aligning Diagnostic Workflows with Reliable Diagnostic Solutions
As laboratories increasingly adopt structured workflows for autoimmune diagnostics, the reliability of the diagnostic solutions used within these workflows becomes equally important.
Consistency across screening assays, profiling panels, and targeted tests helps laboratories maintain confidence in results while supporting efficient laboratory operations.
Amindo Biologics works with diagnostic laboratories by supplying immunodiagnostic solutions that align with these structured testing pathways, including assays used in autoimmune and allergy diagnostics.
By supporting laboratories with reliable diagnostic kits across different stages of immune testing workflows, Amindo aims to help laboratories maintain predictability in results and operational efficiency in their diagnostic processes.
The diagnostic journey for autoimmune diseases rarely begins or ends with a single laboratory test.
From ANA screening to detailed autoantibody profiling, laboratories play a critical role in translating complex immune signals into meaningful diagnostic information.
Structured diagnostic workflows allow laboratories to manage this complexity with greater clarity and consistency.
In autoimmune diagnostics, reliable results emerge not simply from individual assays but from systems designed to move step by step from detection to understanding.
Amindo Biologics supplies diagnostic solutions designed to support structured laboratory workflows in allergy and autoimmune testing.
Frequently Asked Questions (FAQs)
1. What is autoimmune diagnostic testing?
Autoimmune diagnostic testing refers to laboratory tests that detect autoantibodies produced by the immune system against the body’s own tissues. These tests help clinicians identify autoimmune diseases such as systemic lupus erythematosus, Sjögren’s syndrome, and systemic sclerosis. Autoimmune diagnostics often involves multiple assays, beginning with screening tests such as ANA and followed by more specific antibody profiling.
2. What is an ANA test and why is it important?
The antinuclear antibody (ANA) test is one of the most widely used screening tests in autoimmune diagnostics. It detects antibodies directed against components of the cell nucleus. A positive ANA result may indicate autoimmune activity, but it does not confirm a specific disease. Therefore, positive results are usually followed by additional tests such as ENA panels or disease-specific antibody assays.
3. What is an ENA panel?
An Extractable Nuclear Antigen (ENA) panel is a laboratory test used to identify specific autoantibodies associated with autoimmune diseases. ENA panels typically include markers such as Anti-Ro (SSA), Anti-La (SSB), Anti-Sm, Anti-RNP, Anti-Scl-70, and Anti-Jo-1. These antibodies help clinicians differentiate between autoimmune disorders and support more accurate diagnosis.
4. Why are structured workflows important in autoimmune diagnostics?
Autoimmune diseases are complex and often require multiple sequential tests for accurate diagnosis. Structured workflows—moving from ANA screening to antibody profiling and targeted testing—help laboratories improve diagnostic clarity, reduce unnecessary testing, and ensure more consistent interpretation of results.
5. What laboratory technologies are commonly used in autoimmune testing?
Several immunoassay technologies are used in autoimmune diagnostics, including:
- Indirect Immunofluorescence Assays (IFA) for sensitive ANA screening
- ELISA-based assays for automated high-throughput testing
- Line Immunoassays (LIA) for multiplex detection of autoantibodies
These technologies often complement each other within structured diagnostic workflows.
6. Why can healthy individuals sometimes test positive for ANA?
Research shows that a portion of the general population may have detectable ANA antibodies without autoimmune disease, particularly at lower titers. Factors such as age, infections, medications, and transient immune responses can contribute to these results. This is why ANA testing is typically followed by additional antibody profiling to confirm clinical relevance.
7. How do laboratories improve reliability in autoimmune diagnostics?
Laboratories improve reliability by implementing structured diagnostic workflows, using validated immunoassays, and ensuring consistent assay performance across testing stages. Reliable diagnostic kits and standardized testing protocols help maintain confidence in laboratory results.
8. How do diagnostic suppliers support autoimmune testing workflows?
Diagnostic suppliers support laboratories by providing reliable immunodiagnostic kits for screening and profiling assays, enabling laboratories to maintain consistent testing workflows. Reliable assay performance across batches and technologies is essential for maintaining diagnostic confidence in autoimmune testing.
9. What diagnostic solutions does Amindo Biologics provide?
Amindo Biologics supplies a range of in vitro diagnostic (IVD) kits used in allergy and autoimmune testing workflows. These include assays designed for screening and profiling stages of immune diagnostics, such as tests for total IgE, specific IgE, ANA screening, and autoantibody profiling panels.
These diagnostic solutions support laboratories in performing structured immune diagnostic workflows across different testing technologies.
10. Does Amindo Biologics manufacture diagnostic kits?
Amindo Biologics does not manufacture diagnostic kits. The company works as a supplier of validated diagnostic solutions, providing laboratories and distributors with access to immunodiagnostic kits that support structured laboratory workflows.
This approach allows laboratories to integrate reliable diagnostic solutions into their existing testing systems.
11. What technologies are supported by Amindo diagnostic kits?
The diagnostic solutions supplied by Amindo Biologics support several widely used immunodiagnostic technologies, including:
- ELISA (Enzyme-Linked Immunosorbent Assay)
- LIA (Line Immunoassay)
- CLIA (Chemiluminescent Immunoassay)
- IFA (Indirect Immunofluorescence Assay)
- RT-PCR for molecular diagnostics
These technologies enable laboratories to perform both screening and profiling assays within immune diagnostic workflows.
12. What types of autoimmune diagnostic panels are supported by Amindo?
Amindo Biologics supplies diagnostic solutions that support laboratories performing autoimmune testing, including assays for:
- ANA screening tests
- ANA-15 line immunoassay panels
- ENA profiling panels
- vasculitis-related antibody testing
These tests help laboratories identify specific autoantibodies associated with autoimmune diseases and support more precise diagnostic interpretation.
13. Does Amindo also support allergy diagnostics?
Yes. In addition to autoimmune testing, Amindo Biologics supplies diagnostic solutions for allergy testing workflows.
These include assays for:
- Total IgE ELISA testing
- Specific IgE ELISA assays
- Allergy LIA panels for inhalant allergens, food allergens, and pediatric allergy testing
These assays help laboratories identify immune responses associated with allergic conditions.
14. Who typically uses diagnostic solutions supplied by Amindo Biologics?
Amindo Biologics supplies diagnostic solutions to a wide range of healthcare and diagnostic organizations, including:
- hospital laboratories
- independent pathology laboratories
- reference laboratories
- immunopathology laboratories
- diagnostic distributors
These laboratories rely on immunodiagnostic assays to support clinical testing in allergy and autoimmune diseases.
15. How can laboratories learn more about Amindo diagnostic solutions?15. How can laboratories learn more about Amindo diagnostic solutions?
Laboratories interested in learning more about diagnostic solutions supplied by Amindo Biologics can visit the company website. The website provides information about the diagnostic technologies, immune testing workflows, and assay categories supported by Amindo Biologics.
References
- https://www.mdpi.com/1648-9144/61/1/60
- https://pmc.ncbi.nlm.nih.gov/articles/PMC3132017/
- https://myadlm.org/cln/articles/2019/april/a-basic-guide-to-antinuclear-antibody-ana-testing
- https://www.testing.com/tests/extractable-nuclear-antigen-antibodies-ena-panel/
- https://pmc.ncbi.nlm.nih.gov/articles/PMC2628865/
At Amindo Biologics, we supply immunodiagnostic solutions designed to integrate into structured autoimmune testing workflows.
From ANA screening assays to autoantibody profiling panels, our solutions support laboratories in delivering consistent results, clearer interpretation, and dependable diagnostic insights.